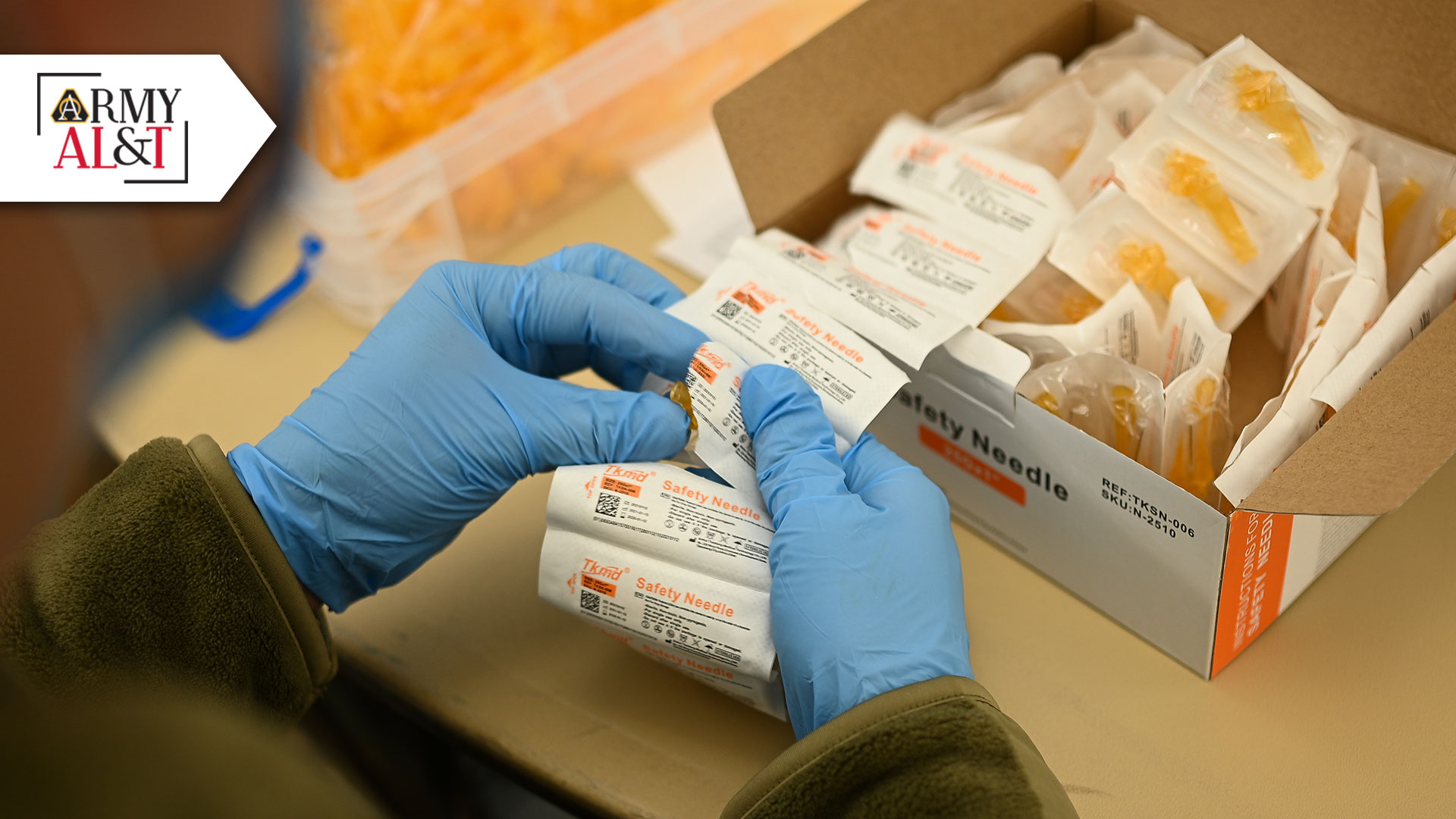
ASSEMBLY LINE: An Airman unpacks safety needles to be used in syringe assembly. Safety syringes and needles have an attached shield to reduce the risk of accidental injury. (Photo by Airman 1st Class Nicholas Rupiper, U.S. Air Force)
U.S. Army Medical Acquisition Team supplies DOD, nation with syringes for COVID-19 vaccinations.
by Erik Heine
A modern syringe is a fairly commonplace item. This small instrument is a simple mechanism that has provided great benefit to mankind. The U.S. military has used it to protect warfighters against biological threats for over 100 years. However, because of the impact of the coronavirus pandemic, the U.S. Army has had to respond rapidly to worldwide demand for this tool in unprecedented ways.
In early 2020, a small team of highly trained medical acquisition professionals—whose motto is “Nevertheless, we deliver!”—was working within the Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense (JPEO-CBRND). It was preparing to supplement the U.S. Army supply of safety needles and syringes to administer COVID-19 vaccinations to Soldiers, once the vaccine was authorized and ready for use. However, a greater challenge loomed on the horizon: ensuring that the necessary needles and syringes would be available to vaccinate the entire nation. While most of the media focused on the efforts necessary to create the vaccines, the underlying goal has always been to deliver those vaccines into the arms of an anxiously waiting public. To do so, the nation needed plenty of safe and effective needles and syringes.
“When the coronavirus hit, we were one of the few who had ever prepared for this type of nightmare,” said Charles Paschal, assistant program manager for JPEO-CBRND’s Joint Project Manager for Chemical, Biological, Radiological and Nuclear Medical (JPM CBRN Medical). “It became clear that the world would be scrambling for needles and syringes on a much larger scale, as soon as the vaccines were discussed.”
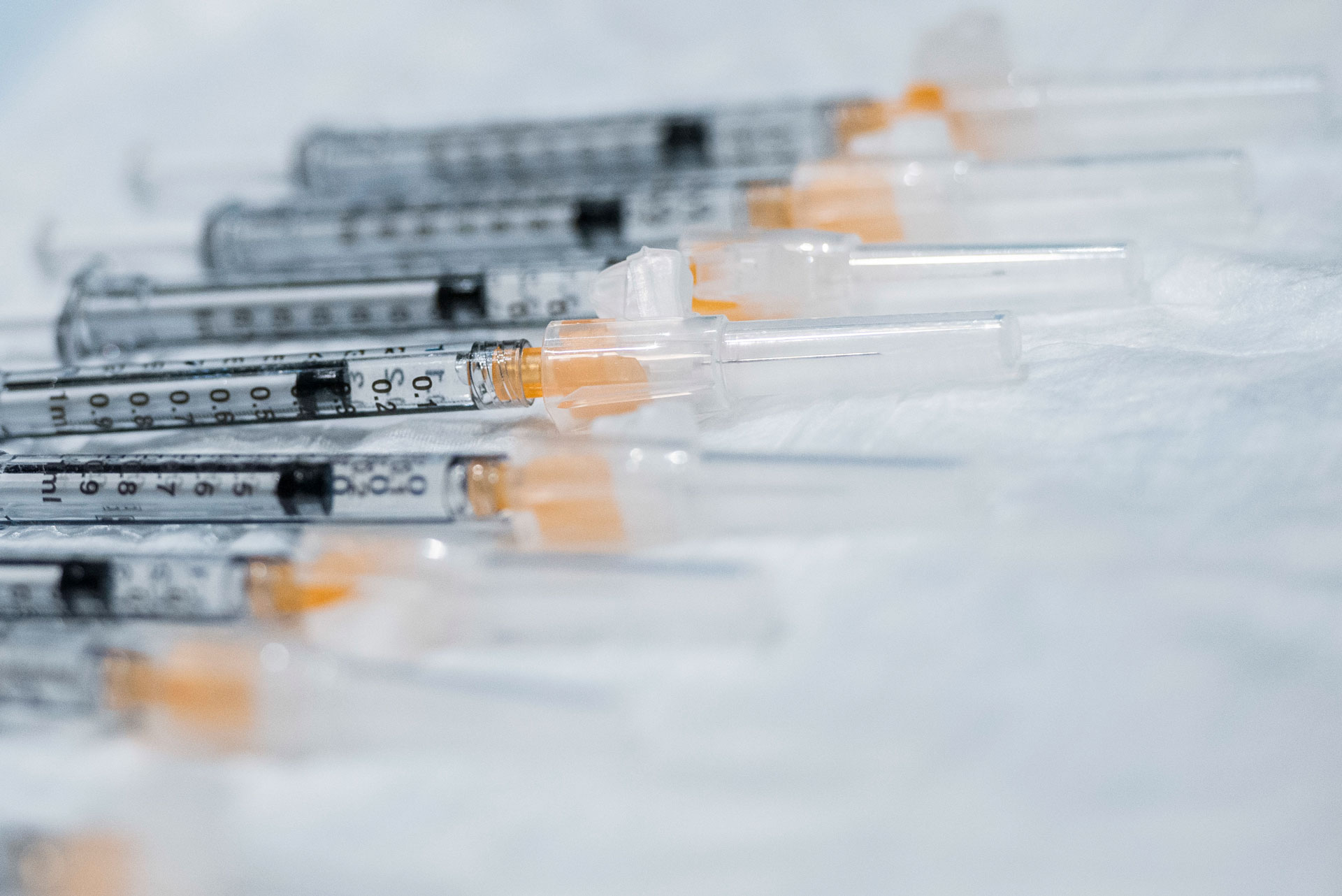
SWIFT RESPONSE: JPEO CBRN Medical provided a total of over 1 billion safety syringes over a 12-month period. (Photo by Wesley Farnsworth, 8th Air Base Wing Public Affairs)
PARTNERSHIPS AND A WHOLE-OF-GOVERNMENT APPROACH
Although the team had never prepared to gather more supplies than DOD needed, it was able to leverage ongoing interagency partnerships in a whole-of-government solution to accelerate the national vaccination campaign, and to rapidly acquire more than 1 billion safety needles and syringes for use throughout the United States. The safety syringes and needles, which feature an attached shield to reduce the risk of accidental injury to health care workers, would be critical to the nation’s COVID-19 vaccination strategy; a challenge of providing a total of over 1 billion safety syringes, across nine contracts, over a 12-month period, with over 88 million planned to be delivered by the end of 2020.
Leading the $310 million, nine-contract effort to ensure that the needles and syringes would be available for the COVID-19 vaccine, was U.S. Army Maj. Andrea Mountney, deputy joint product manager for Chemical Defense Pharmaceuticals at JPM CBRN Medical. “Interagency coordination is critical to successfully executing a mission of this scale with so many moving parts,” said Mountney. “JPM CBRN Medical has operated in the joint space for years, so we were uniquely positioned to leverage those existing relationships and develop new ones. It was a natural extension of what we do every day.”
Mountney led the interagency assisted acquisition to procure and deliver more than 70 percent of the nation’s supply of syringes and safety needles to support the nationwide administration of the COVID-19 vaccine. The national COVID-19 vaccination campaign represented an unprecedented public-private partnership in which development and fielding activities were conducted in parallel, to significantly truncate the schedule. The compressed timeline for vaccine development, as well as additional pressures on an already taxed global medical supply chain, intensified the need for coordination across the government.
As part of the whole-of-government response, the team participated in weekly interagency meetings between representatives from DOD, the Department of Health and Human Services, the Department of State and the Department of Homeland Security. The meetings served as the primary platform to coordinate product shipment, to expedite international and domestic import and export activities, and to synchronize allocation of products across distribution sites for ancillary kit building and nationwide distribution. Ancillary kits typically contain needles, syringes, alcohol pads, vaccination cards and protective equipment for medical professionals, to support approximately 100 vaccinations per kit.
As vaccine candidates emerged and raced toward U.S. Food and Drug Administration (FDA) emergency use authorization, the team proactively pivoted to focus on and prioritize acquiring the low dead space (LDS) syringes that were necessary to draw the maximum number of doses per vial. These syringes were field tested to support drawing six doses from the Pfizer vaccine, prior to FDA approval. The LDS syringes supported a 20 percent increase in the number of U.S. vaccinations, providing over 31 percent of the U.S. government’s LDS syringe procurement goal.

ORGANIZATION STATION: U.S. Air Force Senior Airman Deja Cole, medical technician with the 412th Operational Medical Squadron, 412th Air Base Wing, prepares her station by making a box to hold bandages at the state-led, federally-supported Ford Field Community Vaccination Center in Detroit, April 22, 2021. (Photo by Airman 1st Class Nicholas Rupiper, U.S. Air Force)
ACCELERATING VACCINE ROLLOUT
To support an accelerated vaccine rollout of needle and syringe deliveries for ancillary kits, the team turned to the U.S. Transportation Command (USTRANSCOM), DOD’s premier provider of full-spectrum global mobility solutions. The team modified its transportation requirements across all contracts to accelerate delivery and provide 35 percent more product over the first three months, in support of the emergency use authorization and advanced distribution of the COVID-19 vaccines. While commercial air transportation availability was critically short because of the pandemic, JPM CBRN Medical’s cooperative efforts with USTRANSCOM led to a successful international airlift of approximately 200 million needles and syringes.
Subsequent to the initial COVID-19 vaccination campaign, Mountney and the team worked with counterparts in HHS and supported the U.S. government acquisition strategy for expanded needle and syringe procurements to support four follow-on missions:
- COVID-19 vaccination humanitarian assistance.
- Annual COVID-19 booster shots.
- Support for vaccinations against new COVID variants.
- Replenishment and expansion of the strategic national stockpile inventory.
Together, they helped to develop a multifaceted strategy that leveraged inventory surplus with carefully planned potential new procurement actions in order to maximize product deliveries to meet requirements, while reducing the risk of medical supply chain disruption. Mountney briefed HHS and DOD senior leaders, as well as the White House COVID-19 supply chain coordinator, on the current status of needles and syringes for the COVID-19 vaccination campaign, with focus on the on-hand inventory of ancillary kits and contracted quantities of domestic and foreign needles and syringes.

PACK OUT: Defense Logistics Agency supply technician Brandon Gerber packs Moderna vaccine ancillary kits—which typical contain needles, syringes, vaccine cards and other essential items for 100 vaccines—to be shipped overseas. (Photo by Nutan Chada, Defense Logistics Agency)
“On a daily basis, you just focus on the mission to get the job done,” said Dr. Jason Roos, joint program executive officer for Chemical, Biological, Radiological and Nuclear Defense. “We worked hand-in-glove with several government entities, companies and educational institutions we hadn’t worked with previously, or in a long time. This effort required a lot of teamwork, a lot of hustle, and in many instances, a lot of hours.”
With nearly 150 years of combined professional experience, the JPM CBRN Medical experts are the tip of the spear, facilitating the advanced development and acquisition of medical solutions to combat chemical, biological, radiological, nuclear and emerging threats.
“This team’s track record speaks for itself,” said Col. Ryan Eckmeier, joint project manager for CBRN Medical. “We take pride in our people and the expert knowledge they bring to the table. Time and time again, they have proven that they are the right team to handle something like this monumental task.”
For more information, go to https://www.jpeocbrnd.osd.mil/coronavirus.
ERIK HEINE supports the Joint Project Manager for Chemical, Biological, Radiological and Nuclear Medical (JPM CBRN Medical) at Fort Detrick, Maryland. He is certified as a Project Management Professional and holds a bachelor’s degree in business administration from James Madison University.

