
DECONTAMINATION COMPLETE: The 436th Airlift Wing and the 436th Maintenance Wing complete testing of the Joint Biological Agent Decontamination System (JBADS) Lite on an in-operation C-17. (U.S. Air Force by Roland Balik)
JPEO-CBRND aims its capabilities, medical countermeasures, expertise and acquisition acumen at COVID-19.
by Steven Lusher and Rachel Porto
Those in the chemical, biological, radiological and nuclear (CBRN) community follow the threat mantra, “It’s not if; it’s when.” The rapid global spread of COVID-19 was unprecedented, and the ultimate impact remains unknown. However, the prospect of such an event occurring was not unexpected. History has shown that, whether the threat is natural or manufactured, the best can be done is just to prepare for its eventuality.
The mission of DOD’s Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense (JPEO-CBRND) focuses on protecting the warfighter from America’s adversaries. Its CBRN defense capabilities, medical countermeasures, expertise and acquisition acumen proved vital in accelerating efforts for the U.S. government’s response to the pandemic.
Since 1994, JPEO-CBRND has led the nation’s development of CBRN defense equipment and medical countermeasures to protect the joint force. Working with interagency and industry partners, JPEO-CBRND has developed capabilities for national and international defense initiatives, providing solutions to challenges ranging from support after the Sept. 11, 2001, attacks, to efficient neutralization of 600 metric tons of deadly chemical warfare agents from Syria in 2014. These experiences helped build JPEO-CBRND’s reputation as DOD’s advanced developer for delivering and fielding CBRN defense equipment and medical countermeasures.
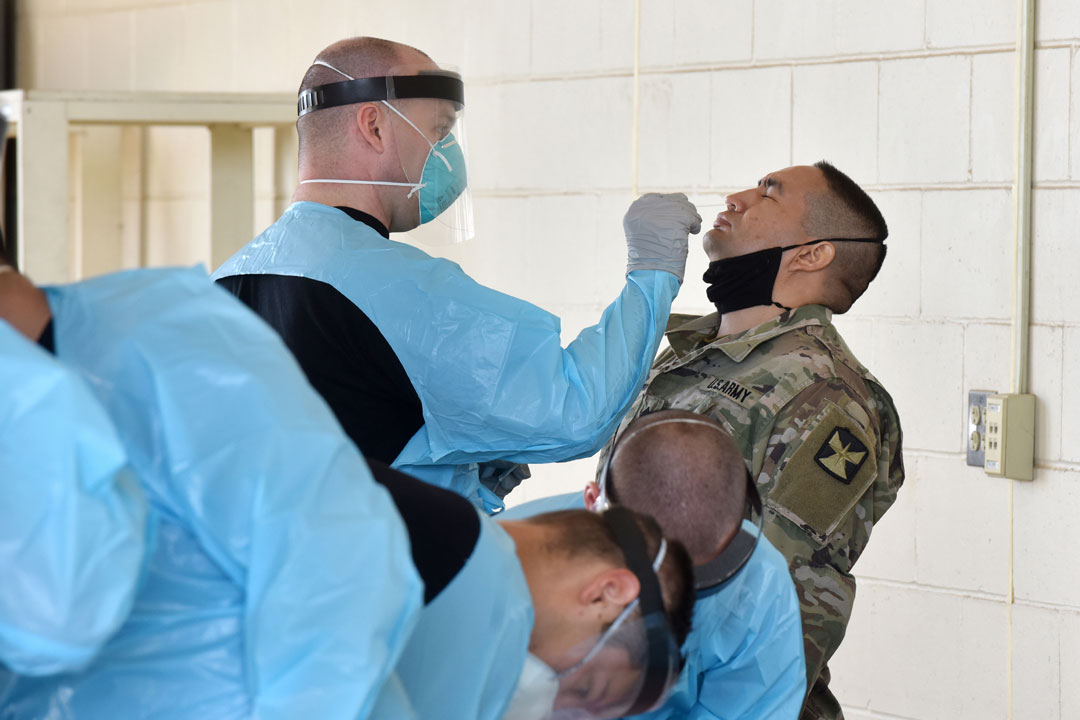
GETTING TESTED: Soldiers are tested for COVID-19 prior to mobilization for training on Camp Ripley, Minn., July 28, 2020. (Photos courtesy of JPEO-CBRND)
Shortly after COVID-19 became a known global threat, the White House’s Operation Warp Speed, an interagency initiative that includes the U.S. Department of Health and Human Services, collaborated with DOD to support development and acquisition of vaccines, diagnostics and therapeutics. This partnership would leverage the CBRN defense and acquisition expertise of the JPEO-CBRND and its existing relationships to find immediate solutions for DOD and the nation. JPEO-CBRND was perfectly positioned to serve as a liaison among the interagency, industry and academic partners, and was central to enabling and managing the largest contract awards in its history at a record-breaking pace.
“By leveraging existing acquisition strategies and equipment, the JPEO-CBRND has helped Operation Warp Speed pave the way for many COVID-19 defense strategies,” said Dr. Jason Roos, deputy joint program executive officer for CBRND. “The JPEO-CBRND’s contributions to the fight against the COVID-19 pandemic have been extraordinary and innovative, and I’m incredibly proud of our team’s tireless hard work in support of Operation Warp Speed.”
Led by the Department of Health and Human Services, Operation Warp Speed had an ambitious goal—to deliver 300 million doses of vaccine by the beginning of 2021. It would require multiple companies racing to find a viable solution to enter through the proper clinical trials for safe and effective use. The operation enlisted experts and support from JPEO-CBRND in the field of vaccine development, biological threats, regulatory matters and acquisition.
In addition to providing support to the White House, JPEO-CBRND worked with the Defense Health Agency (DHA) on DOD support efforts that included the purchase of COVID-19 diagnostic systems and assays, personal protective equipment and medical test equipment as well as conducting therapeutic research.

SUPERVISING PROGRESS: Col. Matthew Jones, 436th Airlift Wing commander and other senior wing leadership stopped by for a short briefing from Maj. Kallander, Ryan Adams and Brant Lagoon as the aircraft and equipment was going through testing. (U.S. Air Force by Roland Balik)
JPEO-CBRND worked with U.S. Army Contracting Command to address Operation Warp Speed and DOD requirements using a combination of Federal Acquisition Regulation (FAR) and non-FAR-based contracts. Together, they executed contract actions quickly and obligated funds within weeks instead of months, while JPEO-CBRND provided total program management, medical and biological expertise, and acquisition support to get the best value for the taxpayer. Within six months, JPEO-CBRND facilitated and managed contract procurements of up to $10.8 billion for Operation Warp Speed, $1 billion in support of DHA, and more than $455 million for DOD. JPEO-CBRND managed the award of these contracts, which were funded with Health and Human Services’ Biomedical Advanced Research and Development Authority and DHA dollars, and mitigated risk by using an existing other-transaction authority mechanism to compress contract negotiations from weeks or months down to days.
This efficiency enabled Operation Warp Speed to accelerate delivery of protective and test equipment and to leverage existing scientific research to enter Phase I clinical trials for vaccine development. Long before the COVID-19 pandemic, JPEO-CBRND used this same other-transaction authority, which allows rapid and direct pre-award negotiations with industry, to partner with innovative and nontraditional companies. As a result, JPEO-CBRND was able to draw on years of in-depth experience with other-transaction authority negotiations and awards in order to accelerate contract actions for COVID-19 efforts without compromising integrity or accountability.
“The DOD has specific skills, authorities and experience to work these types of quick-turn actions,” said George Sfakianoudis, JPEO-CBRND chief financial officer. “In order to apply the DOD’s abilities to support Operation Warp Speed, DHA and others, a thorough review of the contractual and financial systems and processes was done before we executed the first penny against these efforts.”

PRECISE PREPARATION: Erica Borgers-Klonkowski, left, Alice Young, center, and Brant Lagoon prepare conical tubes for testing of the Joint Biological Aircraft Decontamination System Lite on a C-17A Globemaster III. The tubes contained Phi6, a biosafety level 1 bacteriophage. (U.S. Air Force by Roland Balik)
EXISTING CAPABILITIES:
One of JPEO-CBRND’s primary customers is the National Guard Bureau. As the nation’s first line of defense, the National Guard’s weapons of mass destruction civil support teams assist state and local authorities responding to domestic CBRN incidents. The teams are in every state, on standby 24/7 throughout the year. As COVID-19 grew into a global pandemic and the number of infected people increased in the United States, the weapons of mass destruction civil support teams were on the front lines, providing testing capabilities to some of the nation’s hardest-hit areas.
Much-needed personal protective equipment and diagnostic testing became priorities for first responders in the hardest-hit areas. JPEO-CBRND worked with the Defense Health Agency to expedite the procurement of masks, face shields and gowns, and accelerate the fielding of COVID-19 diagnostic systems. In the 2020 fiscal year, JPEO-CBRND delivered more than 58 million items and has procured more than 1 billion items in total. JPEO-CBRND accelerated fielding of DOD’s Next Generation Diagnostic System, known commercially as the BioFire FilmArray, to the joint force and the National Guard Bureau. Because of the need to expand testing quickly, and the understanding that no single system or company could meet the overwhelming demand, JPEO-CBRND also leveraged the Medical CBRN Defense Consortium Other Transition Authority to work with Cepheid Inc. to develop a testing platform that could provide clinical diagnostic test results within 45 minutes, compared with several hours.
The systems that were either put into place, or were already being implemented at JPEO-CBRND to enable rapid response to CBRN threats were used heavily to support the government’s COVID-19 response. These included antibody production, DNA plasmid support and development, access to rapidly discovered monoclonal antibody candidates, and assessment of whether monoclonal antibody candidates were stable and able to be manufactured.
“We leveraged our existing relationships and formed new partnerships, which are critical to our ability to meet our day-to-day mission, to move our COVID-19 support efforts forward as fast as possible,” said Chuck Nuce, JPEO-CBRND chief of operations and international.
The largest of these existing relationships and partnerships is DOD Advanced Development and Manufacturing, a contractor-owned, contractor-operated facility located in Alachua, Florida, designed to develop and manufacture medical countermeasures for the warfighter. The state-of-the-art equipment designed for agile and flexible manufacturing reduces the time and cost of product development.
One of the many reasons for establishing the facility was to ensure that DOD has priority access to a domestic manufacturing capability during crises such as chemical or biological events and emerging infectious disease outbreaks. JPEO-CBRND used the manufacturing technologies inherent to the advanced facility to rapidly manufacture multiple vaccines and antibody candidates for clinical trials and deployment to DOD personnel. Development of the products was in partnership with the National Institutes of Health and the Defense Advanced Research Projects Agency’s Pandemic Prevention Platform teams, which were instrumental in discovering antibodies, screening and choosing lead candidates to transition to the facility for manufacturing to support Phase I and II clinical trials.
Throughout these COVID-19 support efforts, a One Network of Excellence for Regulatory Affairs and Quality Assurance team, a group of regulatory and quality assurance experts, provided expertise for rapid development and deployment of the test capabilities, vaccines and therapeutics. The support of this team was needed to ensure the active management of critical quality parameters during the development process and that development addressed U.S. Food and Drug Administration requirements, lessening the risk of failure.
To strengthen its support for the national COVID-19 response, DOD also drew on the capabilities of the Global Biosurveillance Portal—a web-based data-visualization and decision-support tool established in 2014 for the U.S. Special Operations Command—as well as DOD and other agencies’ personnel to build situational awareness of the coronavirus. The addition of both the Johns Hopkins University and World Health Organization COVID-19 visualization tools provided real-time, up-to-date information to decision-makers, tactical users and action officers. The Global Biosurveillance Portal continues to add users to provide a whole-of-DOD, whole-of-government approach to the pandemic.
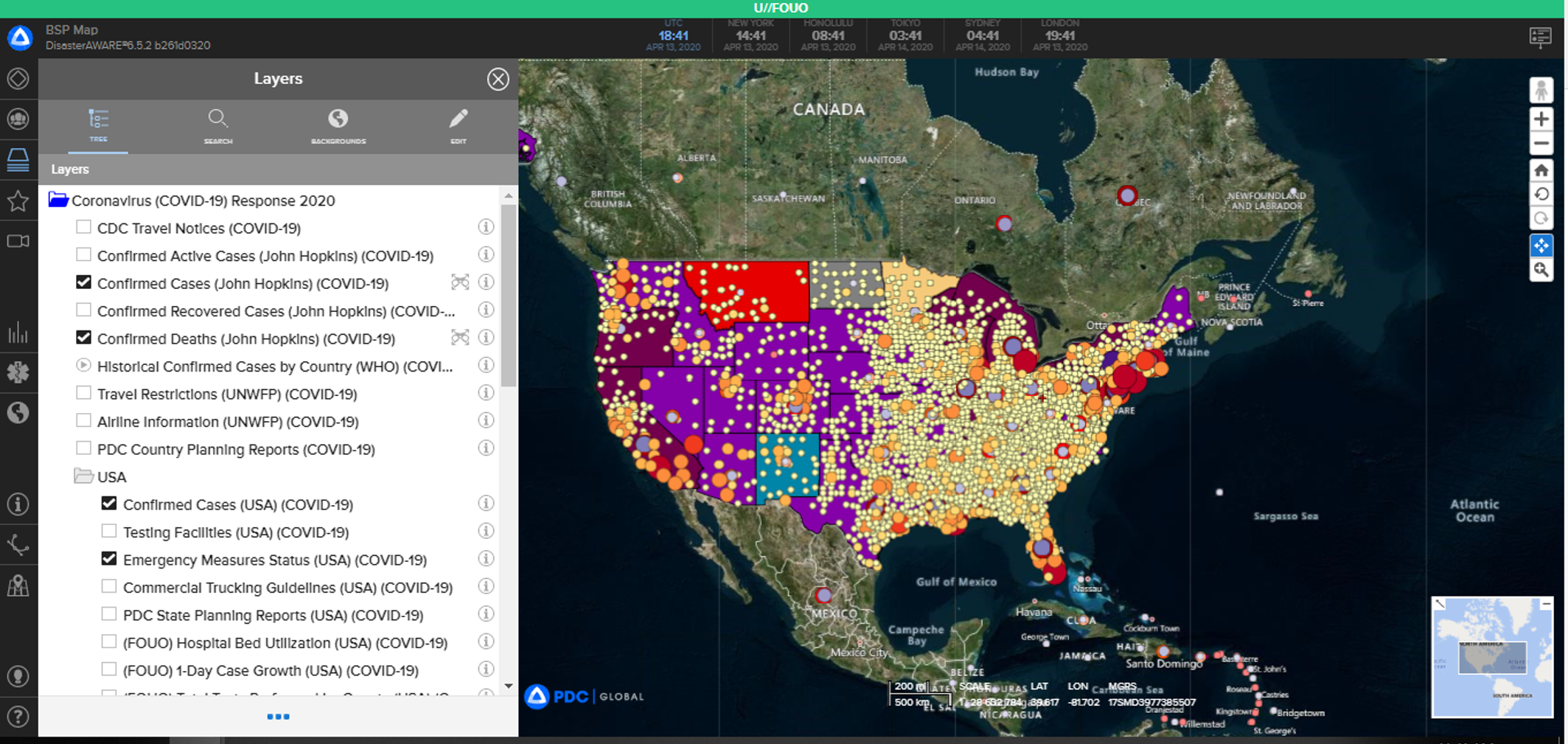
ALL-SEEING EYE: A snapshot of the Global Biosurveilliance Portal, a web-based data-visualization and decision-support tool helps personnel to build situational awareness of the coronavirus. (Photo courtesy of JPEO-CBRND)
THE WAY AHEAD
Developing capabilities that mitigate the impact of current and emerging threats continues to be the primary mission for JPEO-CBRND, even while supporting ongoing COVID-19 efforts. The urgency of the pandemic response required JPEO-CBRND to simultaneously meet that day-to-day mission while working at a record-breaking pace to execute billions of dollars across dozens of contract actions in a matter of weeks to facilitate the nation’s rapid response.
“What we have accomplished in six months is incredible, and is the result of our talented, dedicated and unwavering workforce,” said Douglas Bryce, the joint program executive officer for CBRND. “We continue to fight this battle against COVID-19 and will use the lessons we’ve learned to further develop and procure the best chemical, biological, radiological and nuclear defense equipment for the warfighter.”
While the next CBRN threat is unknown, one thing is certain: The national response, regardless of whether the threat’s origin is natural or nefarious, is largely the same and the systems, platforms, processes and expertise already in place to support JPEO-CBRND’s mission can greatly enhance urgent national efforts if called upon for support, just as they were leveraged extensively to accelerate the nation’s COVID-19 response. Equally important were the strong, well-established interagency and industry partnerships that JPEO-CBRND leaned on to facilitate swift and wide-ranging collaboration and open communication across organizational lines. These relationships, and the contributions they made possible to the full spectrum of the COVID-19 response, will have lasting benefits for the future of CBRN defense for both DOD and the nation.
The nation’s ability to quickly and successfully respond to future CBRN threats, regardless of their origin or intention, requires a thoughtful and critical analysis of the lessons learned from responding to COVID-19. These lessons must inform the development and acquisition of future defense capabilities against these threats. It should also ensure the invaluable relationships developed and strengthened by this crisis are maintained and codified for if and when the next event occurs.

GROWING CULTURE: A lab technician works on a machine used for cell culture growth at the DOD’s Advanced Development and Manufacturing Facility in Alachua, Florida. (Photo courtesy of Ology Bioservices, Inc.)
For more information about JPEO-CBRND’s COVID-19 response, go to https://www.jpeocbrnd.osd.mil/coronavirus or follow JPEO-CBRND on social media at @JPEOCBRND, or contact usarmy.apg.dod-jpeo-cbrnd.mbx.jpeo-cbd-public-affairs-office@mail.mil.
STEVEN LUSHER is the public affairs officer, deputy strategic communications and Freedom of Information Act officer for JPEO-CBRND. He has served on a developmental assignment at the Pentagon for the assistant secretary of the Army for acquisition, logistics and technology and is currently serving on a developmental assignment with the Program Executive Office for Simulation, Training and Instrumentation. Before his position at the JPEO-CBRND, he was a graphic designer for Camber Corp. and served in the U.S. Army as a multimedia specialist supporting the Army Operations Center and the chief of staff of the Army. He has an MBA in marketing and strategic communications from the University of Phoenix, graduated from the Defense Information School for Public Affairs, and is Level II certified in program management.
RACHEL PORTO is a contractor with Kalman and Company Inc. supporting the JPEO-CBRND Strategic Communications Office. Before working with the JPEO-CBRND, she supported various defense efforts as a technical writer and editor. Porto holds an M.S. in professional writing with an emphasis in journalism from Towson University and a B.A. in psychology from the University of Maryland, Baltimore County.
Read the full article in the Winter 2021 issue of Army AL&T magazine.
Subscribe to Army AL&T News – the premier online news source for the Army Acquisition Workforce. ![]() Subscribe
Subscribe

