
CRITICAL EFFORT: The DA2 screening and diagnostics team was able to secure the onshore production of pipette tips. They are critical to molecular-based testing and sustainment of the nation’s blood supply. (iStock image by anyalvanov)
Defense Assisted Acquisition cell cranked up manufacturing to support the fight against the pandemic.
by Erik Heine
In the early months of 2020, as the U.S. government and the nation watched with trepidation as COVID-19 arrived on our shores, “testing” became the buzzword that was at the forefront of everyone’s minds. Unfortunately, the need to test so many people in such a short period of time severely impacted national stockpiles of testing supplies. Supply manufacturers, many of which were abroad, could either not keep up with demand, or could not deliver product because of shipping issues. Among the maelstrom of response activities, Lt. Col. John Nuckols and the Defense Assisted Acquisition (DA2) screening and diagnostics team executed some of the most complex and nuanced medical acquisition efforts both inside and outside of the United States, with vendors who traditionally have not done business with the U.S. government.
|
WHAT’S DA2? The Defense Assisted Acquisition Cell, nested within the Joint Rapid Acquisition Cell in the Office of the Assistant Secretary of Defense for Acquisition, is poised to rapidly respond to the nation’s most urgent acquisition needs in current and future national emergencies. It has assumed the interagency efforts for COVID-19 medical resource acquisition. Leveraging acquisition and subject matter experts from across DOD to meet urgent U.S. government acquisition requirements, it provides policy and oversight to drive awareness and unity of effort. Following DOD’s acquisition support to the Department of Health and Human Services and the Federal Emergency Management Agency during the COVID-19 pandemic, the undersecretary of defense for acquisition and sustainment (USD(A&S)) recognized the need to ensure acquisition assistance. Accordingly, the USD (A&S) established the Defense Assisted Acquisition Cell on Oct. 13, 2020. Built on the urgent capability acquisition that the Joint Rapid Acquisition Cell executes on behalf of DOD during wartime, the DA2 Cell acquires goods and services as rapidly and effectively as possible for the federal government in support of the American public. The Defense Assisted Acquisition Cell provides acquisition assistance when the magnitude of the government’s requirements overwhelms the lead response agencies, leveraging the Economy Act to provide acquisition assistance on behalf of and in coordination with interagency partners. (SOURCE: Defense Assisted Acquisition Cell (DA2) website, https://www.acq.osd.mil/asda/jrac/da2/index.html) |

OVERSEAS DEPLOYMENT: An 82nd Airborne Division paratrooper gets tested for COVID-19 at Fort Bragg, N.C., in May. The testing is in preparation for participation in Swift Response 21, a joint, multinational Airborne exercise involving more than 7,000 paratroops from 10 NATO nations. (Photo by Master Sgt. Alexander Burnett, 82nd Airborne Division)
TESTING, TESTING, TESTING
Early in the pandemic, one of the main issues hindering response efforts concerned overcoming the shortage of specialized specimen collection swabs. From the start, Nuckols and the DA2 screening and diagnostics team were aggressive in their efforts to increase U.S.-based swab production. Initially, they were able to increase the production rate from a baseline of 14.5 million swabs per month (pre-COVID-19) to the potential to produce approximately 147 million swabs per month. However, as the pandemic continued, and additional waves of increased infection occurred, the U.S. Department of Health and Human Services (HHS) realized the national requirement was actually closer to 300 million swabs per month. Performing an exhaustive market search of swab manufacturers across the U.S., the DA2 screening and diagnostics team identified opportunities to increase U.S.-based production of specimen collection swabs optimal for COVID-19 testing, which are expected to provide approximately 350 million swabs per month. This represented a 24-fold monthly increase in the specialized swabs, which are essential to the performance of all U.S. Food and Drug Administration (FDA) emergency use authorization COVID-19 tests in the U.S.
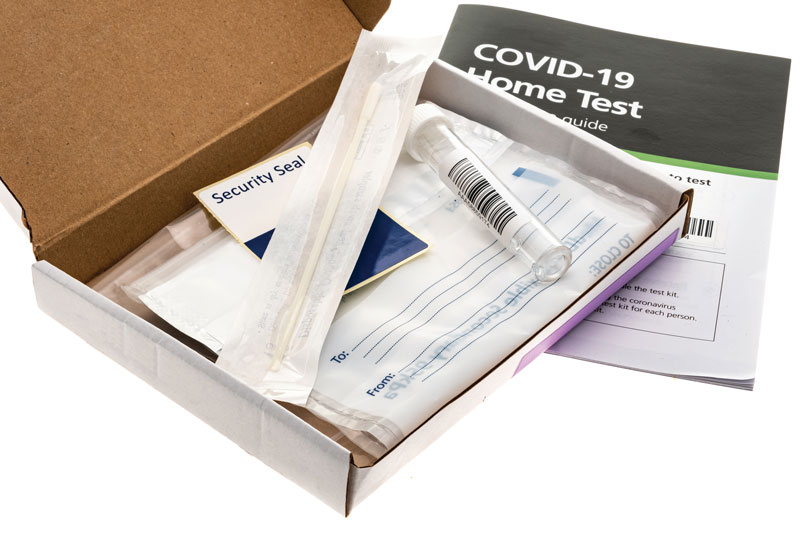
HOME TEST: A COVID-19 home test kit. The DA2 screening and diagnostics team enabled the manufacturing expansion of two at-home testing capabilities that are expected to produce 600,000 at-home tests per day. (iStock image by clubfoto)
The team also invested in the growth of test kit manufacturing on behalf of the HHS Office of the Assistant Secretary for Health, the HHS Office of the Assistant Secretary for Preparedness and Response and the Biomedical Advanced Research and Development Authority. Starting with the clinical laboratory, where most diagnostic tests before and during the pandemic have been performed, the team managed six investments in the expansion of diagnostic test kits and their associated analyzer platforms, expected to increase test kit production by 64 million tests per month. Next, they shifted their focus from laboratory-based testing to the tests performed at the point of care, such as in clinics and doctor’s offices, with an emphasis on testing underserved populations as well as senior citizens in assisted living or retirement communities. This resulted in three investments to increase manufacturing for these point-of-care tests to 10 million tests per month. The team anticipates further increasing production capacity of point-of-care tests by 30 million per month in the near term.
Recognizing that additional testing opportunities existed, the DA2 screening and diagnostics team worked with Office of the Assistant Secretary for Health and representatives from the White House Coronavirus Task Force and the COVID-19 Health Equity Task Force to identify, invest in, expand production of, and procure COVID-19 tests with an FDA emergency use authorization for utilization by individuals from the convenience of their homes. Not only did these investments make access to testing easier in the U.S., they also reduced the potential for exposure of high-risk individuals receiving testing from a clinic, doctor’s office or laboratory. Through their efforts, the team picked up the torch on two research and development investments by the Biomedical Advanced Research and Development Authority and the National Institutes of Health’s Rapid Acceleration of Diagnostics initiative and funded the manufacturing expansion of two at-home testing capabilities, expected to provide the capacity to produce 600,000 at-home tests per day.
The team manages a testing and diagnostics portfolio of $2.1 billion in investments, with an expected $1.1 billion in near-term awards. These investments are expected to yield the tests and testing supplies required by HHS and the nation, including a total expected increase of 120 million tests per month. This represents the majority of the target increase in U.S. testing capability defined by HHS.
BRING IT ON HOME
Another major hurdle during the pandemic revolved around international sourcing of supplies. Between the lack of availability, transportation issues and governmental restrictions, the team understood that onshore production was one of the keys to increasing capacity, effectively slowing the spread of COVID-19. The DA2 screening and diagnostics team received funding from the HHS Office of the Assistant Secretary for Preparedness and Response (ASPR) to build a team of military, civilian and contractor personnel to provide continued industrial base expansion expertise and counsel on acquisition efforts, and to maintain situational awareness regarding availability of industry resources. These integration efforts advanced the development of the Rapid Acceleration of Diagnostics, the Biomedical Advanced Research and Development Authority and the Office of the Assistant Secretary for Health and ASPR initiatives, resolved urgent test distribution crises and aligned industry partners to enable development, and ultimately delivery, of the next diagnostic capability.
As an example of this enhanced interagency collaboration, working with the U.S. Department of State, the team ensured on-time delivery of manufacturing equipment necessary to produce COVID-19 testing materials and brought industry to the table to discuss efforts move their manufacturing to the U.S. in support of COVID-19 testing. Working internationally, and paired with their ongoing U.S. industry engagement activities, the team was able to secure the onshore production of pipette tips, which are critical to molecular-based testing and sustainment of the nation’s blood supply. While onshore production was ramping up, the DA2 screening and diagnostics team further ensured the national supply of pipette tips for COVID-19 testing and blood product testing were never exhausted by coordinating weekly airlifts of 3 million to 6 million pipette tips per week, from approved manufacturing facilities in Germany to distribution centers supplying the nation’s clinical laboratories. This effort became critical to ensuring there was no interruption of highly sensitive molecular testing in the United States.
In addition, the investments in the onshore production of pipette tips was followed by supply chain investments in critical test reagents and other diagnostic supply chain resources. These supply chain investments ensure sustainability of the test expansion efforts made thus far, support the U.S. diagnostic infrastructure and prepare the U.S. medical system and strategic national stockpile for the next pandemic.

MAKING GRADE: Elementary schoolchildren wearing protective face masks in the classroom. The DA2 screening and diagnostics team helped establish a national testing network in support of the national priority to reopen K-8 schools. (iStock photo by kevajefimija)
SCHOOL’S OUT
One of the greatest challenges the DA2 screening and diagnostics team executed, on behalf of and in coordination with HHS, was the effort to establish a national testing network utilizing untapped diagnostic testing resources and laboratory capabilities in support of the national priority to reopen kindergarten through 8th grade (K-8) schools. This investment included execution of three other-transaction authority agreements through Army Contracting Command – Rock Island, Illinois, to establish regional testing hubs across the United States. The DA2 screening and diagnostics team seamlessly aligned HHS’ vision with U.S. Army program management, contracting and technical support resources, soliciting a robust interest and response from the diagnostics industry. The White House COVID-19 test lead deemed this capability essential to reopening K-8 schools immediately; keeping them open in the fall when students return to school, and continued expanded access to testing for underserved populations across the United States.

PASSING THE TEST: Soldiers from the 111th Theater Engineer Brigade medical section conducts COVID-19 testing at Camp Buehring, Kuwait. Every Soldier on Camp Buehring is required to test negative before redeploying to the United States. (Photo by 1st Lt. James Mason, 111th Engineer Brigade)
WALK THIS WAY
Throughout the COVID-19 pandemic, using their acquisition capabilities and acumen, the DA2 screening and development team has always delivered. In addition to being professional acquisition experts, the DA2 team members are “pathfinders,” exploring all solutions and strengthening the resolve of the relationship between DOD and HHS. The team structure they have built is the model adopted by other DOD teams supporting the White House executive order to expand enduring U.S. domestic vaccine capacity, by building and maintaining infrastructure to increase long-term pandemic preparedness. They continue to codify efficiencies and support development of policies and procedures to maintain expedited actions on national priorities, expansion of U.S.-based production capacity, and timely procurement of immediate need testing resources. Their support of the nation throughout this pandemic has moved the country forward, and helped it outlast the adversity of the moment.
For more information, go to https://www.jpeocbrnd.osd.mil/coronavirus.
ERIK HEINE supports the Joint Project Manager for Chemical, Biological, Radiological and Nuclear Medical (JPM CBRN Medical) at Fort Detrick, Maryland. He is a Project Management Professional and holds a BBA in computer information systems and decision support systems from James Madison University.

