
THE COLLECTION: Maj. Juan Guzman, the chief of the Armed Services Blood Bank Center–Pacific Northwest, shows Assistant Secretary of Defense for Health Affairs Thomas McCaffery where the center stores the convalescent plasma it collects. McCaffery visited the center to see the process for collecting the plasma. (Photo courtesy of John Wayne Liston, Madigan Army Medical Center)
A team of seven help hundreds of patients receive COVID-19 treatment.
by Ashley N. Force
The greatest threat to a globally spreading virus is a dedicated team of people working against it. An Army is even better.
The U.S. Army Medical Materiel Development Activity’s (USAMMDA) Force Health Protection (FHP) Division has treated hundreds of COVID-19 patients at dozens of treatment sites on land, on aircraft and at sea.
The FHP Division has an important mission: It provides urgent investigational treatment, prophylactic or diagnostic capability to the warfighter against high-consequence threats when no U.S. Food and Drug Administration (FDA)-approved products or feasible solutions exist. It operates on behalf of the secretary of the Army as lead component, and the surgeon general of the Army under DOD Instruction 6200.02.
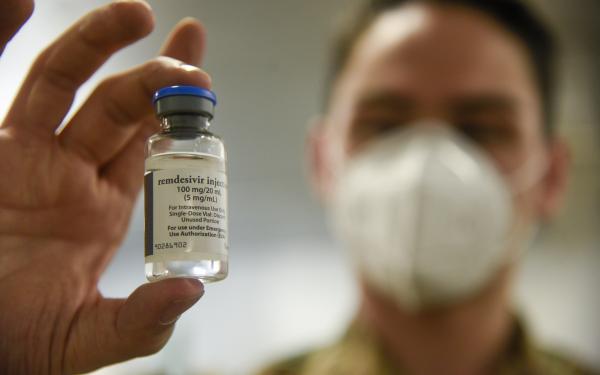
THE TREATMENT: Remdesivir is a broad-spectrum antiviral drug used to treat COVID-19 patients. (Photo courtesy of Force Health Protection Division, USAMMDA)
During the COVID-19 pandemic, FHP, in collaboration with the U.S. Army Medical Research and Development Command (USAMRDC), successfully carried out two treatment protocols and a distribution mission. Its efforts have provided, and continue to provide, additional treatment options to all COVID-19 patients within the Military Health System.
REMDESIVIR EXPANDED ACCESS PROTOCOL
The FHP Division provided the antiviral drug remdesivir to patients diagnosed with COVID-19 as a treatment option. This was accomplished through an expanded access protocol that allowed for early access before the initial COVID-19 clinical trial results. Remdesivir is a broad spectrum antiviral drug that was originally developed as a potential treatment for Ebola virus disease. Researchers found that it may not work perfectly for Ebola, but it showed great activity against COVID-19.
In order to initiate an expanded access treatment, a protocol must first be written, submitted for review, and approved by the USAMRDC Headquarters Institutional Review Board (IRB), as well as the FDA. An investigational new drug application must be filed and approved by the FDA, and then have a number assigned to it. To obtain the product, FHP also must have an agreement in place with the product manufacturer.
“In our case, the product managers of remdesivir were initially under the Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense,” said Army Lt. Col. Sandi Parriott, director of FHP. “They had it in their portfolio for the Ebola virus.”
In discussions with FHP about using remdesivir to treat a different disease, the initial product managers brought up the idea of the treatment against COVID-19. FHP worked with the product managers and the Gilead Life Sciences to put into place a cooperative research and development agreement (CRADA) with Gilead for COVID-19. The CRADA allowed a cross-reference between Gilead’s FDA-issued number for remdesivir that allowed the Army surgeon general’s sponsorship and use of the product under an FDA-approved FHP treatment protocol to DOD personnel worldwide. Gilead provided the 2,200 doses at no cost to the government.
FHP was in talks with Gilead for about three weeks in February 2020. The CRADA was signed on March 5, received FDA approval on March 17, and the product was received from Gilead on the same day. FHP received Institutional Review Board approval on March 18, shipped the product to their first site, and treated their first patient on March 26.
Parriott said remdesivir was chosen as a treatment option because it was shown to have activity against the coronavirus in animal models and in vitro assays. Also, it was the most advanced product identified during market research. It already had a developed safety profile, because it had been used in the Ebola treatment trials in the Democratic Republic of Congo in 2019.
“We knew it was safe, based on the safety profile. We were aware of potential temporary side effects, but it was the most advanced product that was available for a disease we didn’t expect, or have any treatment for,” said Parriott.
Patients who are being treated with remdesivir receive an intravenous injection once a day for five to 10 days, which is determined by the severity of symptoms. During treatment, the physician follows up with the patient to ensure there are no safety issues, providing updates to FHP on clinical progress, laboratory test results, and any potential issues that may be related to the investigational drug Follow-ups for the patients treated with remdesivir continue for up to 29 days post-treatment. FHP’s main goal is to provide an additional treatment option for patients while providing safety and outcome data to the FDA and the product manufacturer.
CONVALESCENT PLASMA EXPANDED ACCESS PROTOCOL
FHP provided an additional expanded access protocol for COVID-19 convalescent plasma, which is used to treat COVID-19 patients with plasma taken from patients who have recovered from the virus. As of November 2020, 65 patients have been treated with convalescent plasma.
Convalescent plasma is plasma taken from patients who have already been infected with and recovered from COVID-19. The plasma contains antibodies that can help temporarily boost the body’s ability to fight coronavirus. The idea to provide this treatment was developed through a collaboration between FHP and the U.S. Army Institute of Surgical Research. This treatment had enough anecdotal usage for other diseases to show that it might have some value by strengthening the immune system against COVID-19. The process of collecting plasma is already licensed and validated for the Armed Services Blood Program. The potential benefit of using convalescent plasma outweighed the low safety risk of transfusion plasma.. FHP submitted its own investigational new drug application and expanded access protocol for convalescent plasma through the FDA for approval. However, before moving a product and processes forward, FHP requires approval from Thomas McCaffery, the assistant secretary of defense for health affairs.
Patients being treated with convalescent plasma receive no more than two units of plasma, depending on the physician’s recommendation. After treatment, the physician follows up with the patient to ensure there are no safety issues related to the product, and provides regular updates on clinical progress and laboratory test results to FHP. Follow-ups for the patients treated with convalescent plasma continue for up to 31 days post-treatment.

WEIGHING DONATIONS; U.S. Naval Hospital Guam Hospitalman Apprentice Rebekah Morrison records the weight of convalescent plasma units collected from Sailors who recovered from COVID-19. (U.S. Navy photo by Jaciyn Matanane, Naval Medical Forces Pacific)
EMERGENCY USE AUTHORIZATION DISTRIBUTION
FHP and USAMRDC’s Office of Regulated Activities were able to get the expanded access protocol for remdesivir, as well as convalescent plasma, written and approved faster than any of their previous expanded access protocols. This resulted in the Defense Health Agency selecting FHP to receive, store, distribute, and track remdesivir under an emergency use authorization.
FHP has managed the distribution of remdesivir to 63 sites worldwide as of November 2020. Before COVID-19, a typical protocol managed by FHP may have included three sites to oversee. At this writing, FHP has already shipped product for treating 648 patients and received 142 resupply requests solely related to COVID-19.
Marianne Erlichman has been working for USAMMDA’s FHP division for 12 years. She has played a major role in coordinating the protocol sites and says FHP has never before managed this many sites and patients simultaneously.
“This has been a learning experience for everybody involved,” said Erlichman. “Whenever we add a site, we have to do an amendment request to the USAMRDC HQ IRB. I am already up to amendment number 39 in just a few months. Normally, after several years, I might be at amendment number eight.”
FHP has treatment sites all around the globe and throughout the United States. Sites range from Japan, Guam, Djibouti, Afghanistan, Kuwait and others. The FHP team has not traveled to the sites, but instead conducted all set-up coordination remotely through phone and email correspondence.
Any DOD-owned military treatment facilities, including aircraft carriers, deployed field hospitals—both inside and outside the continental United States—and hospital ships such as the United States Navy Ship Comfort, can request to be a treatment location by contacting FHP with an inquiry. FHP then exchanges all necessary documents with the group, such as medical licenses and agreements, to become a treatment site. The sites are trained on the protocol and processes, and documentation is then submitted to the USAMRDC HQ IRB for approval. After FHP conducts a site-initiation visit and the product is in place, a patient can be enrolled into one of the treatment protocols.
An approved site can call FHP 24 hours a day, seven days a week in order to provide this treatment option to a patient. Before a patient can begin receiving treatment, the site must send FHP a signed consent form. FHP provides the patient with a special tracking number, which is specific for the protocol that is being activated. After this, the patient can begin treatment. Convalescent plasma for COVID-19 treatment is provided through the Armed Services Blood Program to the active sites, and must also be notified when there is a new patient.
“It’s amazing how many sites we have,” said Erlichman. “We have land-based treatment facilities, on ships and even aircraft carriers, which is a first for us.”
Landstuhl Regional Medical Center (LRMC) in Germany was FHP’s flagship site for both the expanded access for remdesivir and convalescent plasma protocols. Landstuhl has seen a number of patients from numerous locations including the Middle East, Africa, and Europe. It has been treating patients with COVID-19 infections across the spectrum of disease severity.
“We have had very good outcomes thus far with the patients that we have treated,” said Army Lt. Col. Daniel Weinstein, Landstuhl’s chief of internal medicine. “This is likely due to the combination of novel therapeutics, treatment strategies from other diseases that we have incorporated into our treatment plans, and the excellent care that our team provides to all patients that come to LRMC.”
A wide range of patients were able to be treated through the expanded access protocols. These patients include active duty military, dependents, retired personnel, contractors, and in some countries, host nationals.
CONCLUSION
“We want to thank everyone involved in developing and implementing these and other treatments for COVID-19,” said Weinstein. “This is a momentous task that requires efforts from all scopes of practice, research and development. We appreciate it.”
There is one thing that the small yet resilient FHP team is in agreement with, aside from the increasing need for resilience—they are saving lives. They share a mutual satisfaction knowing that, among their efforts, they were able to provide treatment to 753 COVID-19 patients and counting. They have had direct involvement in making these critical treatments available, and in fulfilling their division’s mission.
“I couldn’t ask for a better team—more dedicated, more professional. At two o’clock in the morning, they are taking phone calls, providing customer service, and getting patients enrolled,” said Parriott.
All this is taking place with a team of only seven. Parriott believes that after this experience, her team is ready to handle any future threat of this magnitude.
As Parriott said, “If everyone wasn’t working together as a team, and focused on getting the warfighter treated, we would have never gotten there.”
For more information on the Force Health Protection Division and other projects being developed or managed by the USAMMDA, please visit the USAMMDA website at https://www.usammda.army.mil/.
ASHLEY N. FORCE is functional analyst for the public affairs office at the U.S. Army Medical Materiel Development Activity, responsible for performing research and functional analysis on a variety of projects including event coordination, media coverage, article writing and social media management. She holds a B.A. in journalism from Rowan University. Her journalistic experience stems from working in television news as a general assignment reporter and desk anchor for the CBS-affiliated stations KPIC and KVAL News in Eugene, Oregon.
Read the full article in the Winter 2021 issue of Army AL&T magazine.
Subscribe to Army AL&T News – the premier online news source for the Army Acquisition Workforce. ![]() Subscribe
Subscribe

