
FITS LIKE A GLOVE: Dr. Muthu Wijesundara (left) is fitted with the ReHeal Glove. (Photo provided by University of Texas at Arlington Research Institute)
MTEC gives traction to promising technology for healing Soldier injuries.
by Ramin A. Khalili
After more than two decades as a hand surgeon, Chris Allan was still looking for a solution—a way to solve a frustrating problem. Given that human hands require full, unobstructed motion to function properly, any major surgery on the area usually requires a lengthy and tedious rehabilitation process. Sure, wound care techniques like negative pressure wound therapy are effective, but usage on the hand occasionally comes with just as many drawbacks as benefits. Considering the sheer amount of major hand injuries in the U.S. alone—nearly four million per year, according to Allan—the need for something better was obvious and pressing.
“You do this long enough and you learn that the only way around the mountain is through the mountain,” said Allan, who’s since retired from clinical practice and now works as a researcher at the University of Washington in Seattle, Washington. “So we set off to begin burrowing our way through the mountain.”

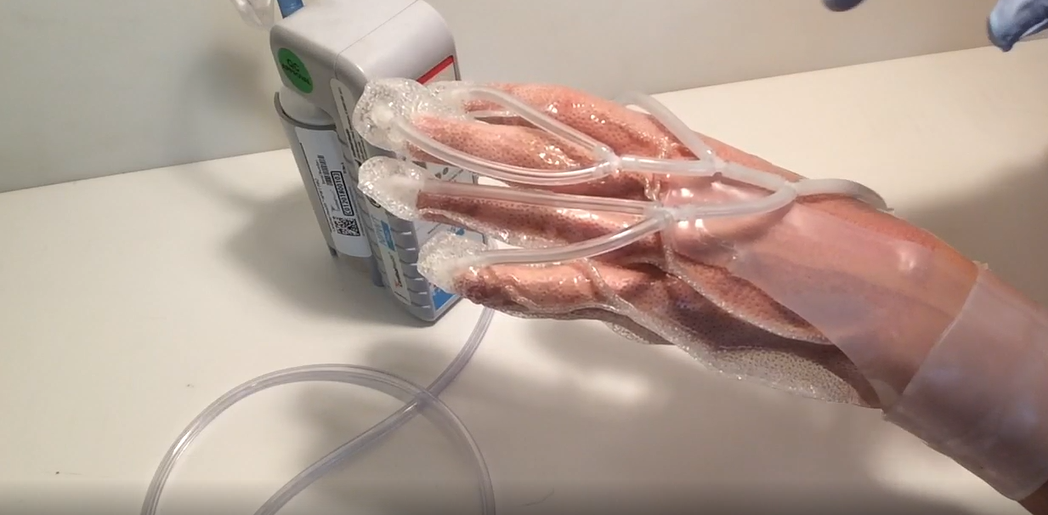
HANDLE WITH CARE: Images of the ReHeal Glove. (Photos by Dr. Muthu Wijesundara, University of Texas at Arlington Research Institute unless otherwise noted)
THE MAGIC GLOVE
All of that burrowing resulted in what is now the ReHeal Glove; a bioengineered glove designed to promote faster healing in surgically-repaired hands. Developed alongside colleague Muthu Wijesundara, a scientist at the University of Texas at Arlington Research Institute (UTARI), the glove is now being tested by the U.S. Army Medical Research and Development Command’s (USAMRDC) Congressionally Directed Medical Research Programs (CDMRP). The military currently sees great promise in a tool with such an effective and portable profile—one with the capacity to help heal a variety of burns and other potential traumatic hand injuries. Indeed, research shows that, collectively, the hands and fingers were the second most common site of non-battle injuries reported by service members during their deployments to Iraq or Afghanistan from 2003-2004; with hand injuries standing as the third most common injury requiring evacuation. According to the same research, 96 percent of those injured were Army Soldiers.
How exactly the ReHeal Glove got to where it is now—flush with funding and undergoing various rounds of testing—is largely due to the role of the Medical Technology Enterprise Consortium (MTEC), a nonprofit corporation that operates as the premier facilitator and collaborator across the entire DOD. Indeed, if it weren’t for MTEC, the near decade-long effort to develop the ReHeal Glove may have withered on the vine. Instead, it provides a promising medical option for millions.
A FLEXIBLE ARRANGEMENT
What makes the ReHeal Glove so unique—the reason it caught the eye of the MTEC team in the first place—is the (literal) flexibility it offers. Patients are able to move their fingers during treatment, a feature that Allan and Wijesundara say reduces the scarring that often results from current rehabilitation techniques that require near-complete immobilization of the hand. Further, the foam dressing used in standard negative wound-pressure therapy treatments—which renders the hand immobile under a vacuum—is eliminated in the glove; replaced instead by clear, flexible, textured silicone, permitting easy application and removal, wound assessment and full range of motion.
For both men, such forward thinking demanded a partner that was on the same page.
“Getting funding for this kind of cutting-edge work is not easy, but MTEC knows this area extremely well,” said Wijesundara, who officially heads the Biomedical Technology Division at UTARI, in reference to the ReHeal Glove’s development process. “Not only does MTEC serve an important role, but they understand the needs for prototype-to-product transition that nobody else wants to fund.”
That explanation of the overall value of MTEC is probably as succinct as it gets; an organization dedicated to fostering collaborations that, in the end, result in moving projects forward. As a 501(c)(3) nonprofit entity, MTEC consists of more than 540 industry and academic organizations committed to developing medical tools that better manage, prevent, diagnose, treat and rehabilitate a wide range of injuries.
MTEC’s consortium-based approach is different from standard funding mechanisms in that it is designed to facilitate frequent interaction between military sponsors, academic institutions, nontraditional defense contractors and large businesses in order to determine exactly what the military is looking for in any particular product.
UP TO THE TEST: The ReHeal Glove during a testing session. (Image provided by University of Washington)
LET’S COLLABORATE
MTEC starts any collaborative effort by conducting outreach; first by scouting through relationships with industrial associations, medical accelerators and venture capital groups, as well as attending scientific conferences and military symposia and judging at company-pitch events—all of which are all instrumental in providing innovation that is ready to meet the military need. This outreach helps capture a large number of those nontraditional partners, thereby constantly bringing new ideas into the military ecosystem.
“In addition, MTEC membership helps innovators get connected with resource capabilities required for prototype maturation and, potentially, with large industrial members that may serve as teaming, co-development or even be potential acquisition partners one day,” said Lauren Palestrini, director of research programs at MTEC. “MTEC has developed strong ties with USAMRDC since its inception in 2015, and our teaming and collaboration capabilities across both university and large and small business members helps our military sponsors access a dynamic, innovative ecosystem that is uniquely suited to encourage small startup companies developing technologies in the military medicine space.”
The tool MTEC uses to get an up-close look at promising efforts is an other-transaction agreement, which is a special contracting mechanism that allows USAMRDC to move forward far more quickly along the contracting pathway than standard Federal Acquisition Regulation (FAR)-based contracts normally allow; a process that in turn increases the speed at which Soldiers receive innovative medical technology. The other-transaction agreement aims to set chosen projects in motion in mere months and allows for complex collaborations, made possible by the mechanism’s reliance on nontraditional partners and built-in flexibility with regard to data rights and regulatory terms. Additionally, other-transaction agreements allow for more conversation and negotiation between the DOD and the performer as compared with traditional FAR-based contracts. ((In this context, “the performer” is the entity developing the prototype. MTEC members can partner with the performer for particular needs.)
With that kind of unique support infrastructure in place, MTEC is able to offer investigators a partnership where, following review and approval of their funding request, they watch that very infrastructure actively work on their behalf; making connections, suggesting teaming arrangements and more.
“We then have competitive methodologies in place that can quickly solicit solutions to the government requirements,” said Kathy Zolman, director of operations for MTEC, describing how the process works. “Then it’s up to the individual performer—whether that’s a university or a company—to propose their [scientific] milestones and associated timelines; and then once on award, we manage and track the project from there.”
Said Palestrini, “I think it’s one of the major benefits of the infrastructure that we’ve built that folks can bring a new technology forward and then have those interactions with the government and with MTEC to help them better refine or steer their technology into a direction that best fits the military need. And concurrently, that technology provider can pursue a broader civilian use case as well.”
GAINING MOMENTUM
This is the terrain Allan and Wijesundara were attempting to navigate back in 2013 when they first began pooling their talents on the ReHeal Glove. Following their successful application for funding, MTEC funded the pair more than $200,000 (via a Prototype-Acceleration Award) for additional research, which—when combined with the one million dollars the team was eventually awarded from CDMRP—catapulted the project into reality. For perspective, the MTEC Prototype Acceleration Award mechanism is instrumental in helping teams advance their prototypes toward their next major technical milestone, with the hopes that additional funding from another source will follow from there. According to MTEC, the award mechanism provides that “awardees may receive up to $250,000 to achieve a technical milestone(s) that helps them secure follow-on funding.”
“I was in an early meeting years ago discussing MTEC,” said Wijesundara. “And I saw very quickly the opportunity that was available, what they had created—the attempt to get semi-mature technology into the hands of clinicians.”
Those attempts have blossomed over the past several years. Since it became fully operational in January 2016, USAMRDC has awarded 176 different prototyping projects through MTEC with more than $640 million in government funding—along with an additional $87 million in cost share funds (which comes via either cash or another type of in-kind contribution by the performer). Currently, MTEC has more than 117 active awards; meaning the opportunity Wijesundara saw all those years ago has made a similar impression across the larger academic and business worlds as well.

PERFECT FIT: Dr. Muthu Wijesundara is fitted with the ReHeal Glove during a lab session at the University of Texas at Arlington Research Institute. (Image provided by University of Texas at Arlington Research Institute)
CONCLUSION
As it stands now, the ReHeal Glove is involved in an early feasibility study clinical trial with the U.S. Food and Drug Administration—a trial funded by CDMRP. The device will be tested on a range of individuals—so far two healthy volunteers have been studied, with injured patients to follow later this year—throughout this process questions have been posed and answered, processes revised and physical changes made before even more testing takes place. This is the life cycle of this kind of technology.
But the promise of MTEC extends far beyond just the initial idea; overall, more than 40 percent of MTEC’s roster of successful projects have secured “follow-on funding”; funding from either the government or the corporate sector that continues to invest in the prototype advancement with the ultimate goal being to turn the device into a final, polished product.
“There are a lot of parties that are just now becoming familiar with MTEC,” said Palestrini. “It’s got this added benefit where everyone—investors, venture capital groups working through our expanded programs for commercialization, industry partners, the foundations we work with—can come into the mix to get a sense of what the government is funding for the military and then, also, what they can piggyback onto for a civilian use case.”
Allan and Wijesundara are hoping for similar success. For Allan specifically, the ReHeal Glove would be the answer to a problem he’s been trying to tackle for his entire career.
“I used to wish for a device like this,” said Allan, firm in his belief the ReHeal Glove could lead to a dramatic improvement in outcomes across the board—including quicker recovery times. “We expect that it’s going to fill a gap that I’ve observed—and also that my colleagues have observed—over the past 20 years.”
For more information about USAMRDC, go to https://mrdc.amedd.army.mil/. To learn more about MTEC, go to https://www.mtec-sc.org. To watch a video about the ReHeal Glove go to https://www.youtube.com/watch?v=N2na9dsvNLY. To learn more about the consortium model, go to https://asc.army.mil/web/news-not-far-at-all/.
RAMIN KHALILI is a writer with USAMRDC’s public affairs office. Before assuming his current role, he spent five years as the knowledge manager for USAMRDC’s Combat Casualty Care Research Program. During his previous work as a broadcast journalist, he earned an Associated Press Award for his work in Phoenix, Arizona, before serving as Chief NASA Correspondent for CBS in Orlando, Florida. He holds a B.A. in communications from Penn State University. He is a frequent contributor to Army AL&T, and an ALTies Award winner. His most recent piece for Army AL&T was “Double Time” in the Spring 2022 issue.

